Funders : UI – PT OneJect Indonesia
Year : 2024
Quantity : IDR 1.108.750.000
Research Member (Architecture Team):
Miktha Farid Alkadri, Raisa Putri Alifa, Heidy Sekardini
Perkembangan Riset
Based on the proposal targets, by December 2024, stages 1–3 as shown in Figure 1 have been completed, with the entire prototype series finalized, including the printing of the prototype. The following is a brief explanation of each research stage.
Stage 1. Planning and Conceptual Design
This stage serves as the foundation for innovation development, involving data collection on user experiences and current nebulizer needs. The process includes identifying needs by analyzing survey results to define specific requirements that the new product must fulfill. Conceptual design development follows, focusing on creating several design alternatives tailored to user needs.

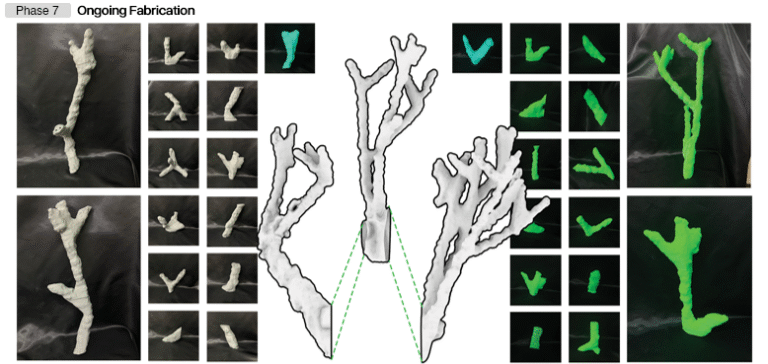
Figure 2. Conceptual design iterations
Stage 2. Prototype Design
In this stage, the best product concept is selected based on an evaluation of proposed designs. The initial prototype is then developed using 3D printing technology to test the feasibility of the design. This stage also involves identifying current nebulizer weaknesses based on survey results, which are then used as evaluation material for the next phase.

Figure 3. Prototype design iterations
Stage 3. Prototype Development
In the prototype development stage, an initial test is conducted to evaluate the prototype’s functionality and its alignment with user needs. This is followed by the production of the final prototype, integrating all components including IoT features. Design refinement is then carried out to address technical issues identified in previous development stages. The final step involves validating functionality by testing all aspects of the product, from mechanical performance to IoT integration.

Figure 4. IoT component assembly
Stage 4. Device Testing
The final prototype is tested to ensure reliability and user comfort. This stage begins with user validation testing through two main activities: a Focus Group Discussion (FGD) with doctors and healthcare professionals as key informants, and a public survey to collect feedback from the target users. The second activity involves satisfaction testing, which measures user satisfaction and acceptance of the product. The stage concludes with data collection and analysis to identify areas for improvement before clinical trials.
Stage 5. Clinical Testing and Evaluation
The final stage focuses on clinical testing and product validation, which includes clinical trials, overall evaluation, and planning for production and commercialization. These three activities are detailed as follows:
- Clinical Testing: Involves healthcare professionals to ensure the product is safe and effective according to medical standards. It also includes testing the efficacy of the nebulizer in delivering liquid supplements.
- Overall Evaluation: Integrates findings from all testing phases to refine the product before entering mass production.
- Production and Commercialization Planning: Develops strategies for manufacturing and launching the product to the market.